Technology
Annexins are calcium 2+ and phospholipid binding proteïns which are evolutionary conserved in a multi-gene family whose members are expressed throughout animal- and plant kingdoms.
In the presence of sufficiently high calcium levels annexin A5 binds with high affinity and specificity to phosphatidylserine in phospholipid membranes. Phosphatidylserine is normally contained on the inside of the cell but it can become exposed (transiently) to the external milieu under stress conditions or when a cell is dying. The original concept of using labeled annexin A5 for detection of Apoptosis (a form of regulated cell death) and the subsequent development of a high affinity assay for detection of Apoptosis are a result from the work of Prof. Dr. Chris Reutelingsperger at Maastricht University, The Netherlands.
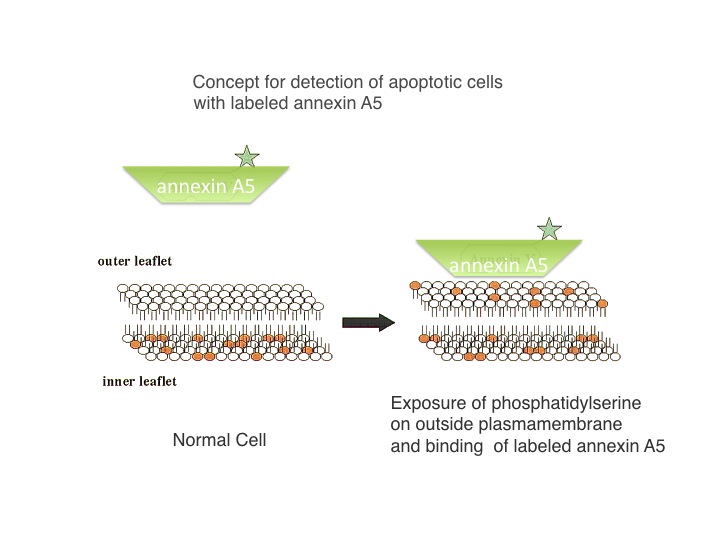
A schematic representation of a phospholipid bilayer with Labeled annexin A5 binding to externally exposed phosphatidylserine (orange colour).
Cys annexin A5
Cys annexin A5 is an annexin A5 that has been designed with a single Cysteine on its concave side, which is opposite to the convex side where the annexin binds to the phospholipid surface. This design allows with thiol chemistry highly specific labeling of the annexin in a 1:1 stoichiometry. The potential for labeling on the convex binding site is eliminated in the Cys annexin A5 which yields a highly pure labeled product that is chemically uniquely defined.
Annexin A5-M1234
Both, wild type annexin A5 and Cys annexin A5, have been engineered in such a way that the ability to bind to PS is lost while other properties of their PS- binding counterparts are conserved. The M1234 versions may be for example be used as negative controls in experiments.